3D printing model for ECM morphogenesis
Extracellular matrix (ECM) is formed outside of producing cells and the mechanism of its morphogenesis remains mysterious. We observed that the surface-tethering and subsequent release of alpha-tectorin/TECTA, a GPI-anchored collagen binding protein, play a critical role in the organization of the tectorial membrane, an apical ECM in the cochlea. We proposed a 3D printing model for the organization of complex ECMs.
However, the model does not explain how distinct matrix shapes are printed from the same cell at different developmental stages and in different locations. How is the specific shape and structure of the matrix organized outside the producing cells? This is a fundamental question in biology since the proper organization of the ECM is critical not only for tectorial membrane morphogenesis but also for tissue morphogenesis in general. We study how the different release modes of the surface organizer regulate matrix patterning.
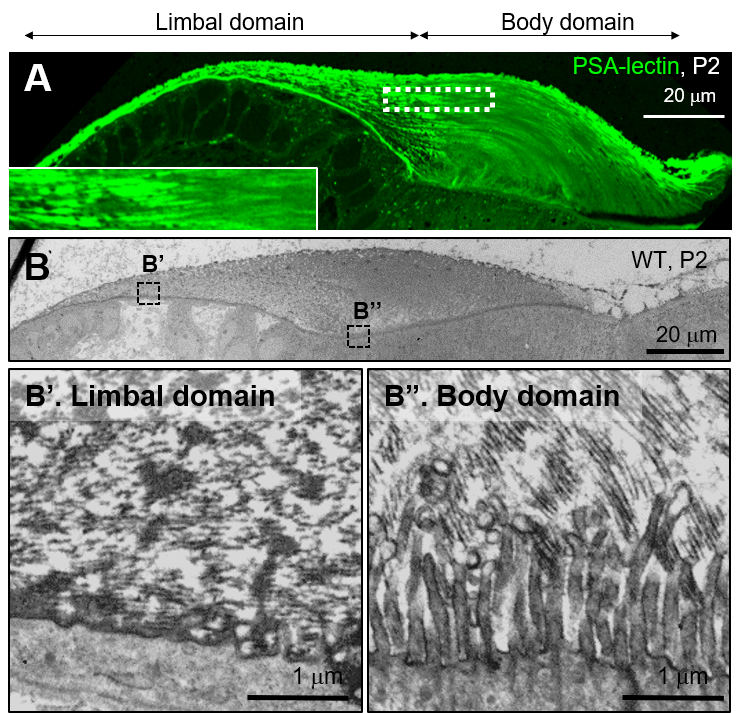
In vivo ECM birthdating
The tectorial membrane is a multi-layered structure. However, the
mechanism by which newly-synthesized tectorial membrane components are incorporated into the previously-assembled
layers is unknown. This is a key question of ECM morphogenesis during
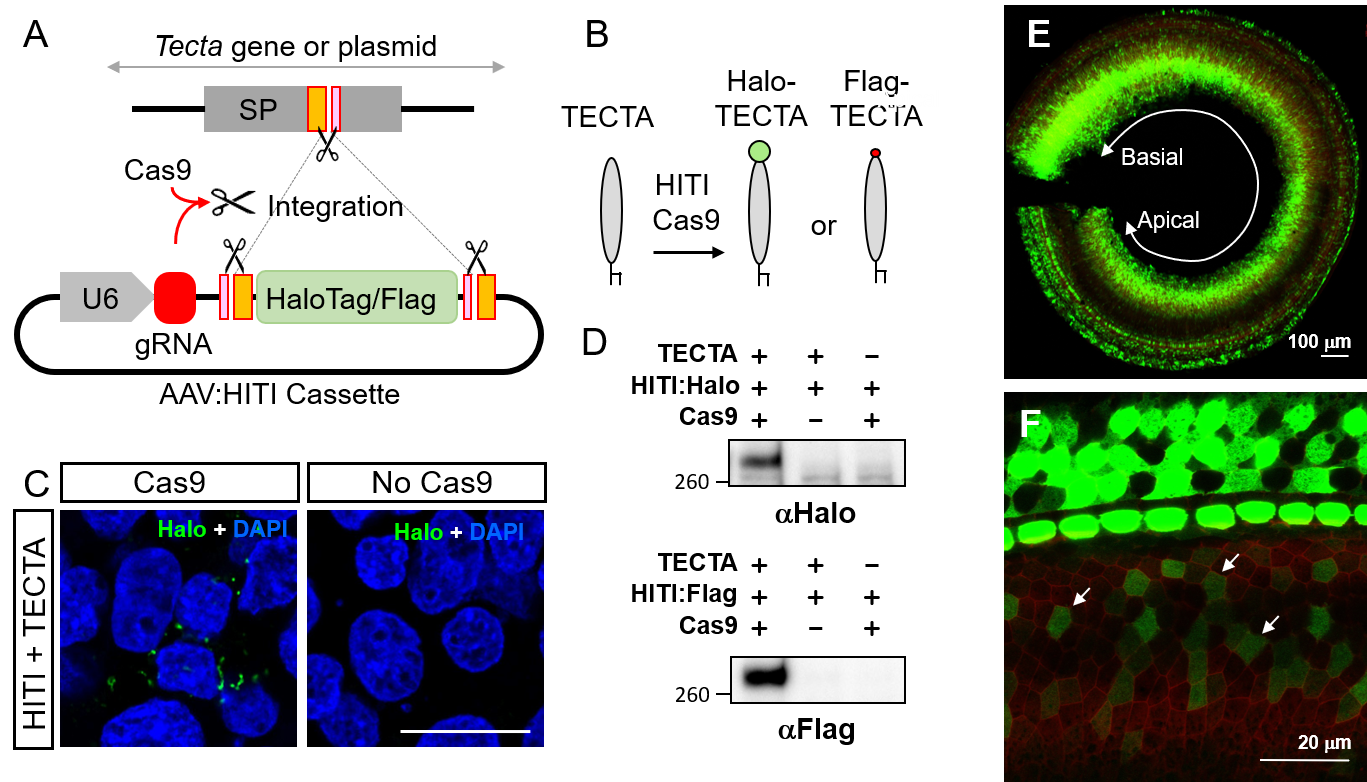
development as well as in a repair process such as wound healing. To visualize the dynamic process underlying matrix elongation, we are developing a novel in vivo ECM birthdating assay by using CRISPR/Cas9-mediated homology-independent targeted integration (HITI) and followed by pulse-chasing of ECM components. Characterizing
the assembly sequence of the multi-layered matrix will advance our
understanding of the morphogenesis process during development and repair
process.
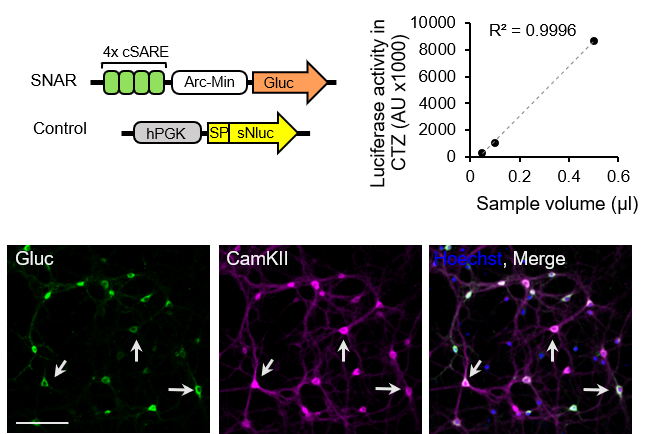
High-throughput activity reporter in live neurons
Neurological and neurodevelopmental disorders are
prevalent worldwide. Despite significant advances in our understanding of synapse
formation and function, developing effective therapeutics remains challenging,
in part due to the lack of simple and robust high-throughput screening assays
of neuronal activity. We developed a simple biochemical assay that allows
for repeated measurements of neuronal activity in a cell type-specific manner. We named this live cell activity assay SNAR (Secreted Neuronal Activity Reporter). By using this novel assay, we can identify neuronal intrinsic and extrinsic factors that modulate synapse development and function in normal and disease conditions. .
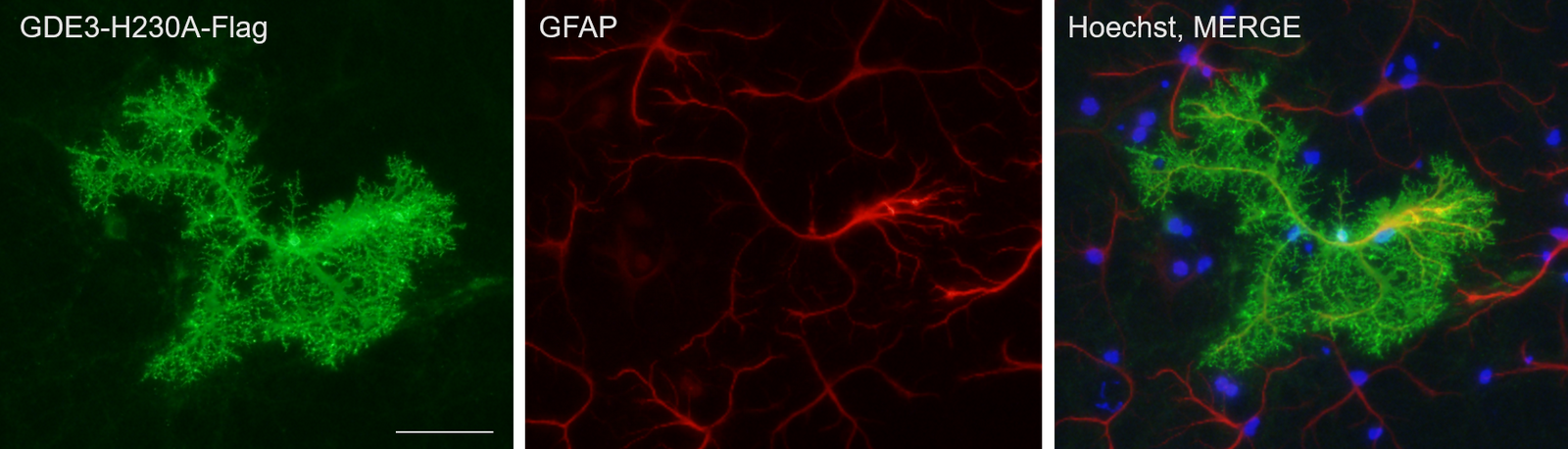
Regulated release of synaptogenic factors from astrocytes
Astrocyte-derived
diffusible factors regulate synapse development and function. However, the
regulatory mechanism underlying the release of astrocyte-derived factors is
poorly understood. Unlike many other secreted factors that do not require a releasing enzyme on the astrocyte surface, the release of GPI-anchored synaptogenic factors can be dynamically regulated by various release modes. We characterize the release mechanism of
astrocyte-derived factors and identify the releasing enzymes and their regulatory signaling.